
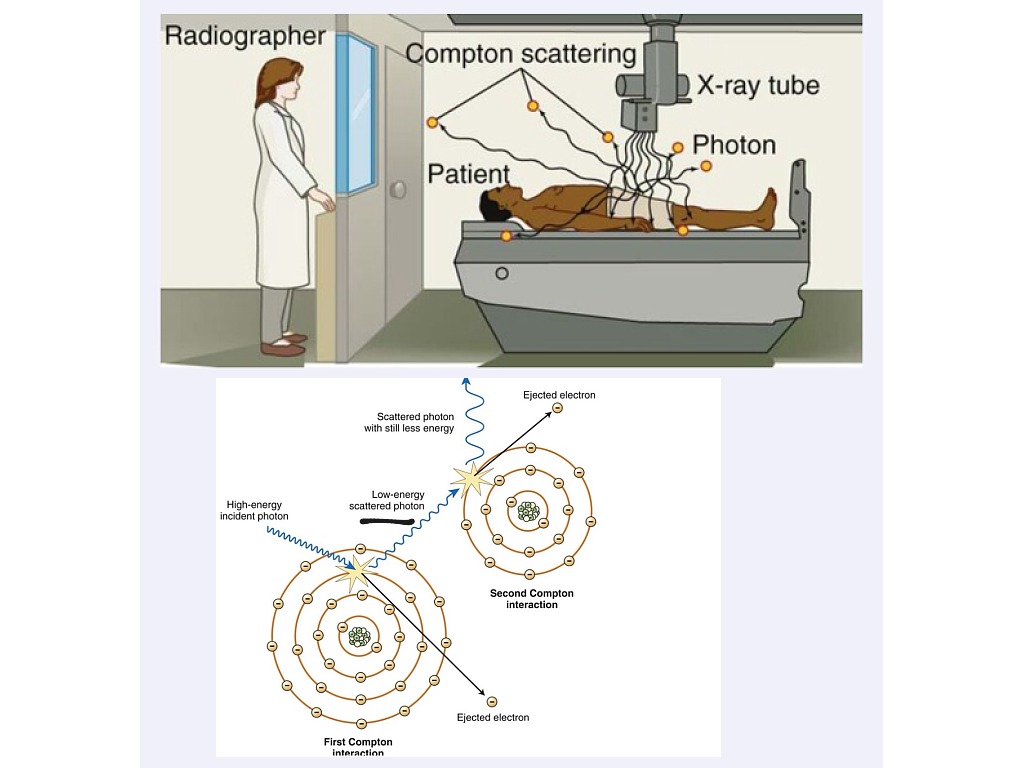
The situation is somewhat similar to a billiard-ball collision and we use the angle, θ, to express the extent of the scattering process. We refer to the ejected electron as a Recoil Electron. 3.2: The Compton Effect - (a) photon scattering and electron ejection leading to (b) an ionized atom. However, in low atomic number materials, such as soft tissues, all atomic electrons can be considered to be essentially free because the binding energy of all electron shells is less than 1 keV. This outer-shell electron is considered to be essentially free of the atom because its binding energy is so low, which is the case in high atomic number materials. Here we can see an incoming X-ray which glances off and ejects an outer-shell electron. It is a radiation scattering event where the incoming X-ray gets scattered by an outer atomic electron - as illustrated in Figure 3.2. Compton Effect The second process of interest is called the Compton Effect. However it can also be inferred that the lower kV will increase the patient absorbed dose since the Photoelectric Effect results in the total absorption of the energy of the incoming X-ray photon. It can therefore be inferred that contrast between bone and tissue in radiographs should increase at lower kilo-voltages (kV). However the extent of this difference is also seen to be inversely proportional to the X-ray energy, so that the absorption difference between bone and tissue is greatly reduced. It can also be used to amplify absorption differences between soft tissues. Since its probability is proportional to Z 3, the absorption in bone, for instance, is amplified relative to that in the surrounding tissue. 
The Photoelectric Effect is of fundamental importance in diagnostic radiography since it is the primary method by which contrast is developed in radiographs. We can therefore infer that the probability of the Photoelectric Effect is approximately dependent on: This probability is very approximately proportional to the cube of the atomic number, Z. the greater the atomic number of the atom, the greater the probability of the photoelectric effect.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
